About Us

Our goal is to bring new therapy to cancer’s most frequently mutated oncogenic target.
KRAS Inhibitor Discovery Strategy:
Focus on Potency, Durable OFF and ON State Inhibition, and Selectivity
Precise. Swift. Lethal.
Exploiting oncogene addiction has been the foundation of extraordinarily fruitful efforts to precisely drug mutated cancer targets and eliminate the malignant cells that carry them. At Kestrel Therapeutics our aspiration is to extend the methodologies and successes of this field to targeting KRAS and then to other significant targets. We have created a foundation of unique science that employs a physical understanding of the drug-target interaction, funded by thought-leading investors, to build a lead program designed to potently address all KRAS mutations in all tumor types. The prospect of patient benefit through the application of these approaches is profound.
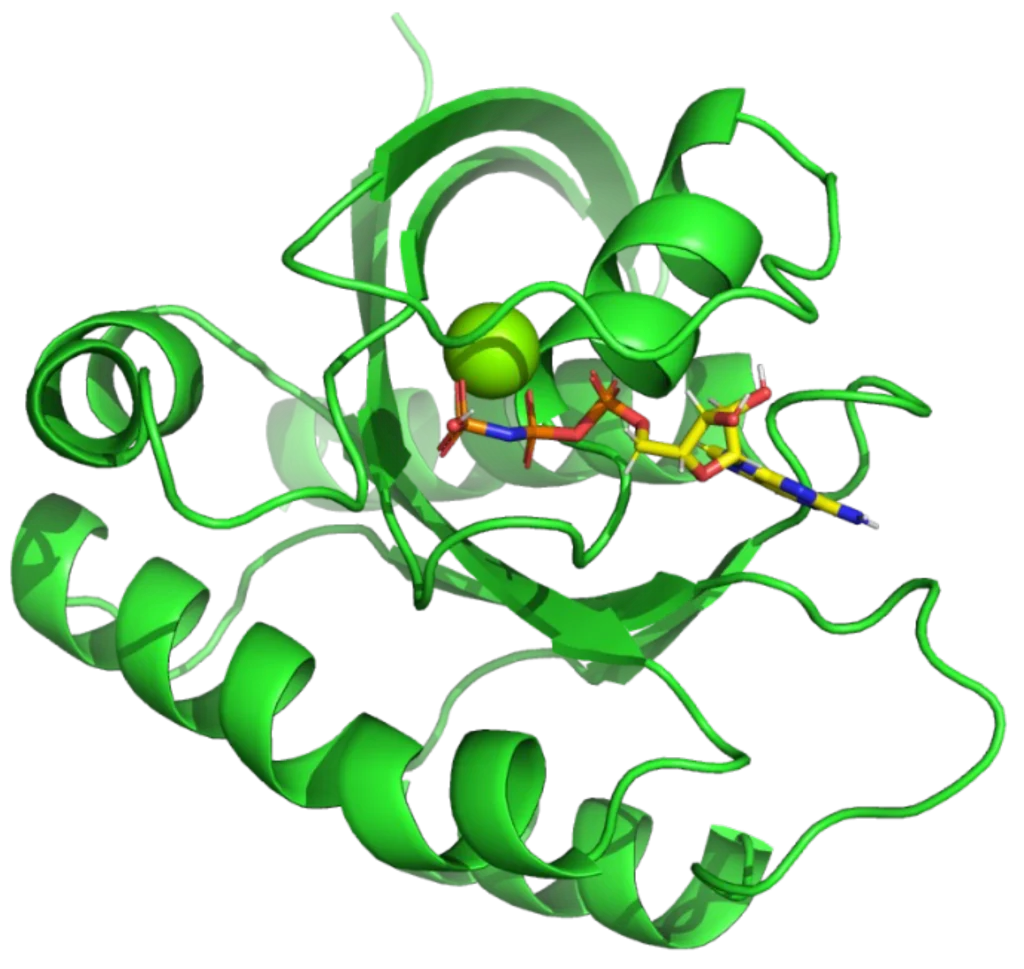
GMPPNP bound to KRAS G12D
Leadership Team
Our experienced leadership team has a proven track record of combining innovation and entrepreneurship to bring transformative technologies to the world of healthcare.

Frank Haluska, MD, PHD
Founder, President & CEO

Mark Bilodeau, PHD
SVP, Chief Scientific Officer

Ashwin Gollerkeri, MD
SVP, Chief Medical Officer

Ross Pettit
SVP, Chief Development Officer

Tom Phair
Chief Financial Officer

Ron Knickerbocker, PHd
SVP, Biometrics & Clinical Development Strategy

Aleksandra Franovic, PHD
VP, Head of Translational Research & Medicine

Omar Ahmad, PhD
VP, Head of Chemical Sciences & DMPK

Shyam Karki, PhD
VP, Head of CMC
Board of Directors
Denis Patrick, PhD
Omar H. Khalil
Andrew Phillips, PhD
Nexo Therapeutics
Nick A. Saccomano, PhD
Barbara Sosnowski, PHD
Frank G. Haluska, MD, PhD
Casey Cunningham, MD
(observer)
Christopher O'Donnell, PHD (observer)
Scientific Advisory Board
Andrew Aguirre, MD, PhD
D. Ross Camidge, MD, PhD
Sharon Campbell, PhD
Channing Der, PhD
Julian Downward, PHD
William C. Hahn, MD, PHD
1Globocan 2022 citation.
2SEER citation.
3US NCI citation.